Translate
Monday, October 10, 2022
Receiving Liquid Hydrogen
Sunday, October 2, 2022
Cryogenic Systems Modeling and Analysis
 |
| Cryogenic Fluid Management (CryoFM™) Interactive Calculations Notebook |
Last month I participated in NASA's annual Thermal and Fluids Analysis Workshop (TFAWS) as a short course instructor, panelist, and presenter on the topic of cryogenic fluid management. This is a critical topic for launch vehicles and spacecraft. It is also becoming a very important consideration for the rapid growth in production, energy storage, ground transportation, shipping, and aviation applications of liquid hydrogen systems.
What is Cryogenic Fluid Management?
- Computational fluid dynamics (CFD): The highest fidelity option that also generally requires the highest level of resource commitment (i.e., computational, personnel, and licensing). CFD typically uses a very fine mesh of finite volumes to model the system. Setting up the model and the appropriate parameter adjustments requires experience with the particular CFD software being used and an understanding of how to best represent the actual system of interest.
- Multi-nodal models: A moderate fidelity and resource option that divides a cryogenic system into discrete lumped nodes. The number of nodes can be few or many, and is a key determinant of the model resolution. Similar to CFD, the modeler's experience with the software and ability to accurately represent the actual system is critical.
- System-level and first-order analysis: The lowest fidelity option with generally the lowest resource commitment. Reduced order system models and first order analyses can be used early in the development to narrow the trade space of feasible designs. Also useful as a check on the results obtained from higher fidelity tools.
System-Level and First-Order Analyses
- How can all of the planned cryogenic fluid management tools best be developed and maintained?
- What about users who don't have access to the tool platforms or aren't permitted by their organization to download them (e.g., Python)?
- If other platforms are of interest to specific users (e.g. Matlab), who will modify the tools for those users?
The Open Source Option
Sunday, July 24, 2022
Transferring Liquid Hydrogen
Pressurant Gas Injection
Autogenous Pressurization
Pumps and Bladders
What About Delivery?
Sunday, July 10, 2022
Storing Liquid Hydrogen
 |
| Composite cryogenic demonstration tank (courtesy NASA) |
Single-Wall Tanks
Double-Wall Dewars
Penetrations
- Supports, flanges, ports and similar structural components
- Fill line for loading LH2 into the vessel
- Drain line for removing LH2 (a single fill/drain line is sometimes used)
- Feed line for high LH2 consumption rate applications
- Pressurization or other pressure building subsystem
- Vent line for pressure relief and fluid conditioning (sometimes tied into the pressurization line with appropriate isolation valving)
- Sensors for temperature, pressure, and mass gauging or fill level monitoring
Reference
Saturday, June 25, 2022
Entering the Cryo Zone with Hydrogen
Liquefying Hydrogen
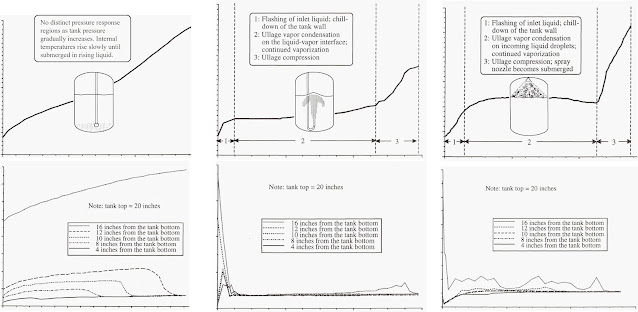
Liquid Hydrogen Storage Behavior
Cryogenic Material Properties
- Yield and ultimate strength: generally increases at lower temperatures for most solids
- Ductility: some materials remain ductile (e.g. aluminum alloys, austenitic stainless steel with > 7% nickel, most face-centered cubic metals); while some materials become brittle (carbon steel, most plastics, most body-centered cubic metals)
- Elastic modulus: varies
- Fatigue strength: varies
References
Sunday, June 12, 2022
The Coming of Age for Liquid Hydrogen Systems: 1960 - present
 |
| Apollo fuel cell (left); shuttle external tank (middle); new LH2 dewar tank at KSC under construction |
Large Scale Liquid Hydrogen Systems Deployment
Hydrogen Fuel Cells
References
Sunday, June 5, 2022
The Evolution of Hydrogen Systems: 1930 - 1960
Liquid Hydrogen
Aircraft Flight Testing with LH2
Beyond Aircraft
References
Sunday, May 22, 2022
Energy Shapeshifting with Hydrogen
 |
| Day operation HyERA™ example system model: renewable power is greater than demand yielding hydrogen production and storage |
Energy Carrier
Energy Storage
Universal Fuel
Potable Water
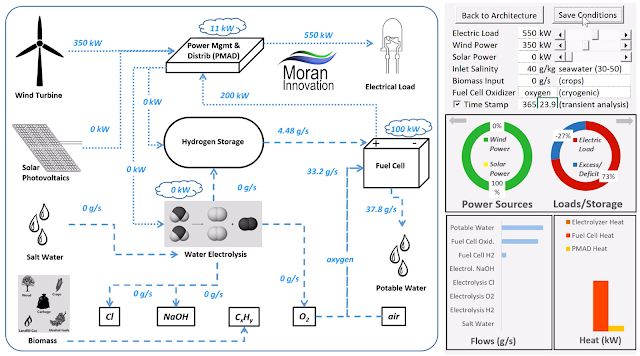 |
| Night operation HyERA™ example system model: renewable power is less than demand requiring hydrogen usage and producing potable water |
Friday, May 13, 2022
The Nuclear Option for Hydrogen
RTGs, NTPs, NEPs, and Fission Surface Power for Space Missions
NIMBY Everywhere
Nuclear Power for Hydrogen Production
Friday, May 6, 2022
Hydrogen is Best Served Cold
Liquefied Hydrogen
- Relatively high volumetric density at low operating pressures
- Long history of production, storage, transport, and usage
- Off-the-shelf subsystems and components available
- Many legacy stationary, over-the-road, train, barge, and launch systems
- Zero boil-off possible with proven cryo-refrigeration technology
- Low temperature provides ancillary system capabilities
Sunday, May 1, 2022
Hydrogen Myth Busting (Episode 2)
Myth #5: Making Hydrogen From Renewables Isn't Practical
- There aren't enough renewable resources to support all the carbon-free hydrogen production needed
- Renewables should only be used to directly meet the electrical demand; producing hydrogen with them is wasteful
- Conclusions regarding hydrogen production from renewables specific to a particular region are globally true everywhere
Myth #6: Hydrogen Isn't Green
- Since most hydrogen has historically been produced with steam methane reforming (SMR), it isn't a solution to climate change
- Hydrogen may contribute to global warming
If hydrogen were to be produced in the quantities required to replace fossil fuels; and large aggregate leaks of hydrogen were permitted to occur throughout this new global hydrogen infrastructure; and furthermore, these leaked quantities of hydrogen managed to reach the upper atmosphere without already combining with oxygen in the lower atmosphere or water; it may combine with hydroxides in the upper atmosphere to form water vapor.
So what's the issue? Keep reading...
This process may inhibit the amount of upper atmosphere hydroxides available to react with the large amounts of leaked methane; thereby inhibiting the ability to mitigate the impact of methane sources and leaks.
While methane is a very potent and prevalent greenhouse gas, it's rather difficult to overlook the circular argument of this scenario. Namely, that transitioning to hydrogen might inhibit the upper atmosphere mechanism that helps to mitigate the greenhouse gas effects of one of the fossil fuels that hydrogen will replace.
The study goes on to recommend that implementation of global hydrogen infrastructures should address leakage to ensure very little reaches the upper atmosphere. The methods and technologies for minimizing hydrogen leaks are well known within the hydrogen community and are already used in any appropriately designed system.
This is a key takeaway and valid consideration as we transition to hydrogen. Unfortunately, that valuable nugget rarely seems to make its way into the subsequent articles that misrepresent the findings. Instead, a more sensationalized tale is spun about the effects of large scale hydrogen usage and greenhouse gases. While that may draw a lot of readers, click-throughs, and online traffic, it is clearly misleading.